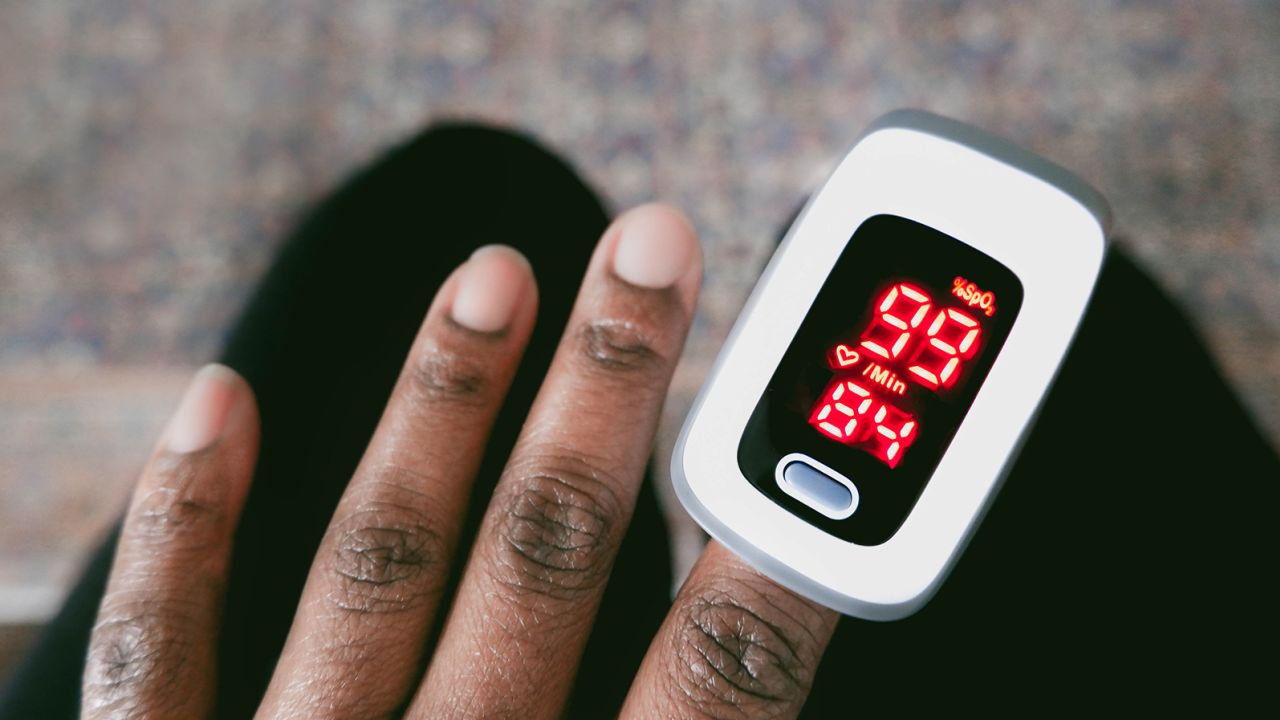
As an emergency medicine physician, Dr. Owais Durrani sees this issue regularly first-hand: When he clamps a pulse oximeter onto a patient’s fingertip to measure their blood oxygen levels, the small device can yield in people with darker skin tones, reporting levels higher than what they actually are.
The possibility of an inaccurate oxygen reading for Black and brown patients worries Durrani in the emergency room on a daily basis.
“Was there a point where I may have discharged a patient that had a reading of, say, 95%, but the actual reading was lower and they maybe could have benefited from observation or being admitted to the hospital? Possibly, and that scares me, and that frustrates me,” he said. “So that’s why it’s so important that the FDA is looking into this.”

ministration’s Medical Devices Advisory Committee darker skin and to discuss recommendations on using these devices on people with dark skin tones and whether they should have labels – such as a black box warning – noting that inaccurate readings may be associated with skin color.
Because the public continues to use these devices to check oxygen levels, “I think we should give more detailed instructions on what they’re supposed to do with them,” Dr. Murad Alam, a member of the panel and professor at Northwestern University, said in the meeting.
“I understand we don’t know exactly what their accuracy is, but I would suggest that FDA might want to consider a labeling change,” he said. “In the meantime, it would allow the FDA the ability to investigate these products and give consumers some guidance as to how to use them – and have some power to actually, at the very least, compel the manufacturers to convey that simplified and expanded labeling to patients who will in fact be using these to try to save their own lives.”
The meeting was intended to help inform the FDA’, their accuracy and performance, and what can be done to fix racial biases. The Anesthesiology and Respiratory Therapy Devices Panel heard from patients, regulators, researchers and developers of medical devices.
“The performance of medical devices needs to be well understood to mitigate any negative, unintended consequences for patients in different populations,” Adm. Richardae Araojo, FDA associate commissioner for minority health, said during the meeting. “So today’s discussion on the real-world performance of pulse oximeters and factors that impact their accuracy will inform FDA as we consider the regulation of these medical devices.”
A ‘critical issue’
Pulse oximeters work by sending light through your finger; a sensor on the other side of the device receives this light and uses it to detect the color of your blood. Bright red blood is highly oxygenated, while blue or purplish blood is less so. If the device isn’t calibrated for darker skin tones, the pigmentation of the skin could affect how the light is absorbed by the sensor, leading to flawed oxygen readings.
Ultimately, the pulse oximeter can estimate the amount of oxygen in the blood without the need for a blood sample. But on a person with dark skin, the oximeter could indicate that oxygen levels are normal, suggesting that the person may be discharged – when, in reality, a blood sample might show low oxygen levels, suggesting that they need not only additional care but oxygen support.

To compensate for the possible discrepancy, Durrani said, he sometimes conducts a “walking” pulse oximeter reading on patients with darker skin.
“So having a patient walk for like five minutes while having a pulse ox on, and usually if they are at higher risk, what will happen is, their oxygen numbers might drop further,” said Durrani, who practices at a hospital in Houston.
A walking test can help him gauge whether the standard pulse oximeter reading overestimated a person’s oxygen levels, but he is still waiting for the day when he can trust the device to provide an accurate oxygen level measurement for all of his patients – no matter the color of their skin.
“I’m definitely very happy that this critical issue is being addressed on a national level at the FDA,” said Durrani, who is not involved in the meeting or with the agency.
“But as an ER doc who’s in the trenches and making a lot of fast-paced decisions based on this data, I’d like to see more discussion hopefully in this meeting or future meetings around what the clinical implications of this are going to be,” he added. “We would need a more kind of multipronged guidance when it comes to how physicians should then interpret these results.”